PUMA System™
The First and Only FDA Cleared Superelastic Fixation Device That Maintains Continuous Compression Without Creep
Indicated for Syndesmosis Fixation, Hallux Valgus Reconstruction, and Tarsometatarsal Fixation.
Available only in the United States.
The PUMA System™ is a nitinol-based fixation device for ankle syndesmosis which provides stabilization without over-compression or loosening with cyclical loading. Nitinol has a proven safety record in foot and ankle surgical repair and reconstruction implants. Bringing nitinol into use for ankle syndesmotic injuries with the PUMA System is a novel innovation with significant advantages over existing screw or endobutton type fixation devices. The PUMA System provides significant syndesmotic stabilization while also allowing for more normal ankle biomechanics. The biomechanical and clinical advantages of this implant have led to improved patient outcomes after these ankle injuries.
Syndesmosis repair by Kent Ellington, MD
“I have used the PUMA System for almost two years in a wide range of patients, from athletes to high-energy trauma, and have seen excellent outcomes with no complications”
Lapidus case by Jason Nowak, DPM, FACFAS
“I highly recommend the PUMA System from Panther Orthopedics for any syndesmotic, lisfranc, or Lapidus stabilization procedure”
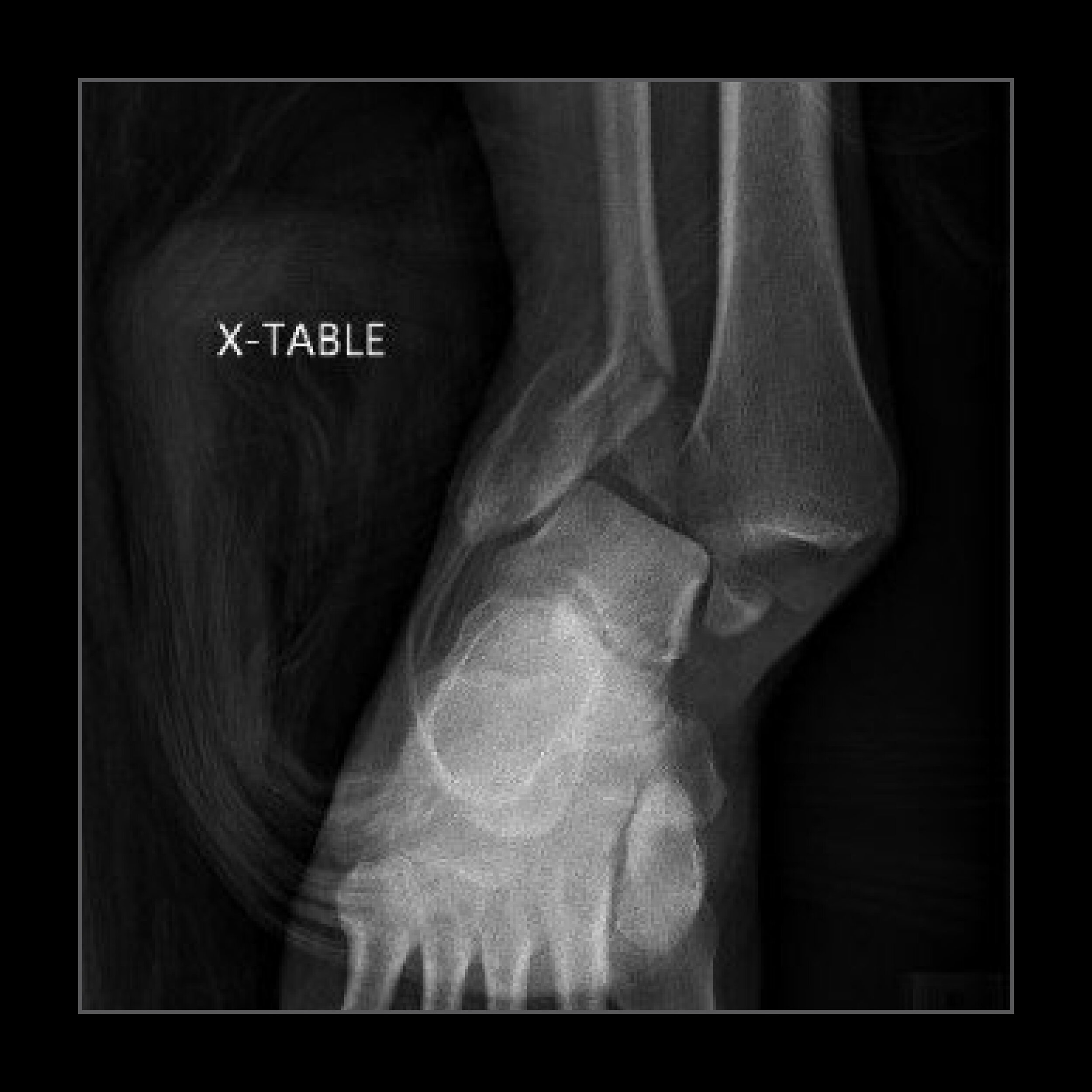
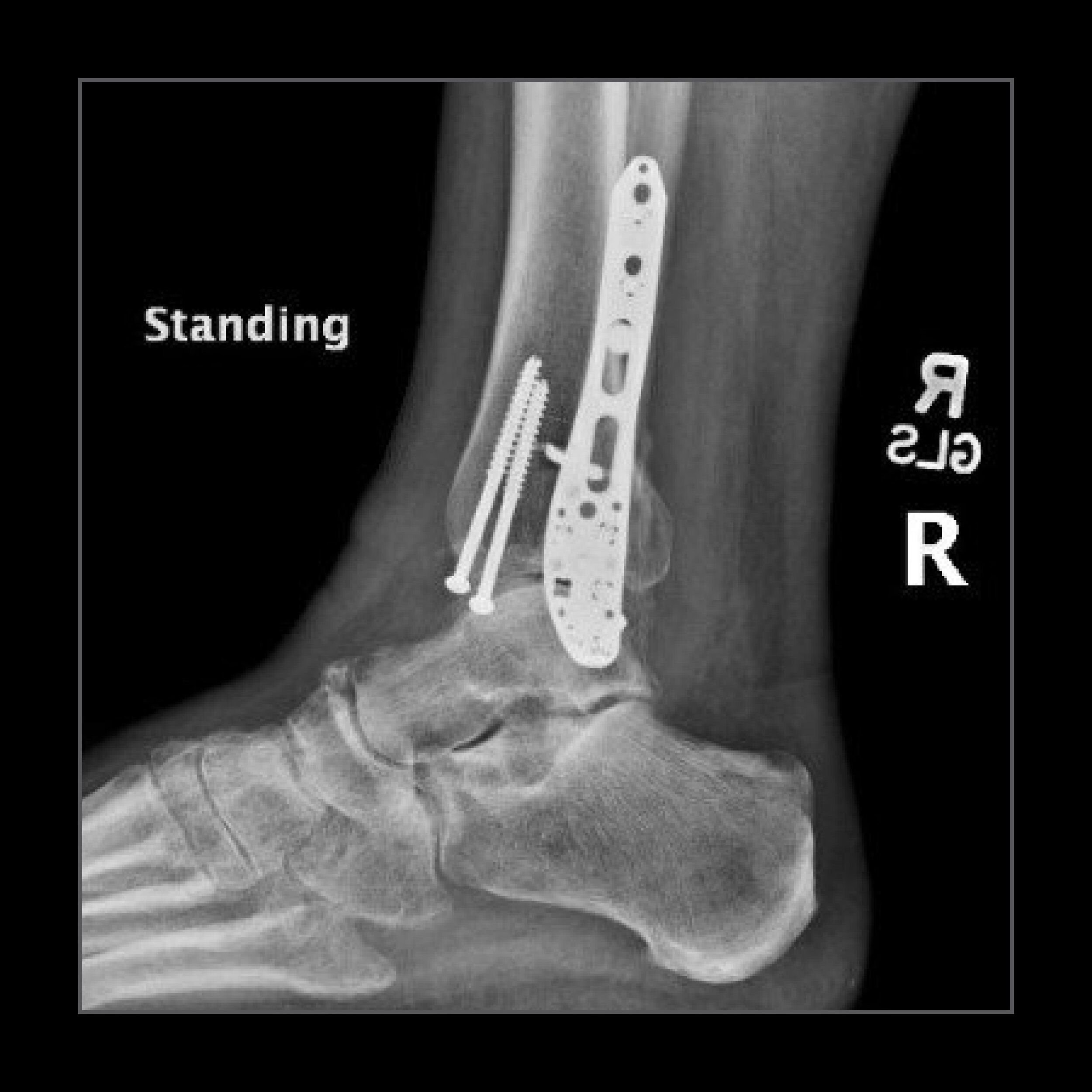
"The patient sustained this bad injury from falling off of a ladder. The patient is doing remarkably well with no activity restrictions. The first x-ray show the original injury, and the other two x-rays show the patient at three months post-op."
Edward Tang, MD
Contra Costa Regional Medical Center
Martinez, California
Successful Mid-Term Clinical Outcomes
We are pleased to announce the commercial success with exceptional mid-term clinical outcomes of the PUMA System
The PUMA System is a soft tissue platform technology
“I consider the PUMA System to be a game changer. Its novel technology allows me to provide the best care available to my patients. Its biomechanics marry perfectly with biology, stability, fixation and strength. I have used it for almost two years in a wide range of patients, from athletes to high-energy trauma, and have seen excellent outcomes with no complications.”
Kent Ellington, MD
Orthopedic Surgeon, OrthoCarolina
Charlotte, NC, United States
Our Story
We are developing superelastic fixation devices for orthopedic trauma and sports medicine applications. Our first product, the PUMA System, is available only in the United States and it is indicated for Syndesmosis Fixation, Hallux Valgus Reconstruction, and Tarsometatarsal Fixation.